Explain the Similarities and the Differences Between Monomers and Polymers
Answer 1 of 2. The smaller molecules are called monomers.
Difference Between Homopolymer And Heteropolymer Definition Synthesis Properties Examples
Carbohydrates lipids proteins and nucleic acids.

. Monomers are small molecules mostly organic that can join with other similar molecules to form very large molecules or polymers. Monomers are small molecules mostly organic that can join with other similar molecules to form very large molecules or polymers. The polymer is more than the sum of its parts.
Monomers are the building blocks of polymers. The same way that links are like a chain or bricks are like a house. Monomers are small molecules in the microscopic scale which cannot be compared to the macroscopic properties of polymers and.
In laymens terms all plastics are polymers but not all polymers are plastics. As amino acids are the monomer units which are combined to form proteins. Monomers are single units that act as the building blocks of polymers.
Answer 1 of 4. A molecule of low molecular weight which may combine with other molecules to form a molecule in a chain or branched form having high molecular weight. Polymers have no such restrictions.
Monomers must have two similar or different functional groups. On the other hand plastics comprise long chains of polymers composed of tiny uniform molecules. In comparison the molecular weight of Polymer is more than a monomer.
These monomers have either double bonds or at least two functional groups per molecule. Hi Grant Youve probably already covered this and I suspect this may be a question from a homework or classwork. What is the difference between a molecule and monomer.
A polymer is formed by polymerization the joining of many monomer molecules. The polymers formed are. Polymers are macromolecules with very high molecular weights built from many repetitive units called monomers.
Vinylic plastics are formed from monomers having a vinyl group. This means they are both made of the same atomic elements however the dynamics and mechanics of. Describe the main function of carbohydrates.
Monomers are simple molecules with low molecular weights. The major difference between polymer and plastic is that polymers exist organically or are created synthetically and comprise joined chains of individual molecules or monomers. Ad Over 27000 video lessons and other resources youre guaranteed to find what you need.
By-products such as ammonia water and HCl are produced. This is an important advantage in the maintenance of cellular osmotic conditions. Compare the processes of dehydration synthesis and hydrolysis.
Is that molecule is chemistry the smallest particle of a specific element or compound that retains the chemical properties of that element or compound. Polymers are large molecular structures made of many monomers. Bonding two molecules together to make a larger molecule by removing water is dehydration synthesis while adding water to split.
Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. A monomer can have different combination units. Monomers are simple molecules with low molecular weight.
The connection between monomers and polymers is that the existence and form of polymers depend on the monomers. Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. Furthermore monomers have a small molecular weight while the polymers have a larger molecular weight which is many times the weight of a monomer.
Addition of monomers results in. Explain the relationship between monomers and polymers. Monomers are small molecules.
Organic chemistry A long or larger molecule consisting of a chain or network of many repeating units formed by chemically bonding together many identical or similar small molecules called monomers. How are polymers formed. Polymers are chains with an unspecified number of monomeric units.
It acquires new characteristics and leads to an osmotic pressure that is much lower than that formed by its ingredients. Explain differences between a monosaccharide disaccharide and polysaccharide. Monomers are small molecules which may be joined together in a repeating fashion to form more complex molecule referred to as polymers.
A polymer is a type of macromolecule that is composed of a large number of repeating units. Polymers are very large molecules made when many smaller molecules join together end to end. A material consisting of such polymer molecules.
Monomer is a word made of two parts mono means one and mer means unit so monomers are the building units of the polymersPoly means many. A monomer can have different combination units. How are they broken down.
Draw a structure of each and explain how each one can be used within organisms. Monomers have similarities in terms of their composition structure and molecular weight. Macromolecules are made up of single units known as monomers that are joined by covalent bonds to form larger polymers.
The basic conceptual building unit of a polymer. And polymers also have more complexity. The key difference between polymer and monomer is that a polymer is a collection of a large number of molecules whereas monomer is a single molecule.
Assuming its ok to answer Ill take a stab at it Quite simply a monomer is a single unit of a many-unit strand of molecules called a. Polymers are complex molecules with very high molecular weight. Polymers are chains with an unspecified number of monomeric units.
Two or more atoms held together by chemical bonds while monomer is chemistry a relatively small molecule which can be covalently bonded to. Monomers must have either a double bond or triple bond. These repeating units represent monomers from which the polymer is made.
Explain the difference between monomers and polymers. Macromolecules are big molecules macro means big opposite to micro which is small.

Difference Between Monomer And Polymer With Table Ask Any Difference

Monomers Polymers Overview Examples What Are Monomers Polymers Video Lesson Transcript Study Com
Difference Between Monomer And Polymer

Difference Between Free Radical And Ionic Polymerization Compare The Difference Between Similar Terms
What Is The Difference Between A Polymer Molecule And A Monomer Are The Monomers Joined By Intermolecular Forces Or Covalent Bonds Are Monomers The Same As The Repeating Units That Make Up

Monomer Vs Polymer What S The Difference Osborne Industries

Question What Is The Difference Between Monomers And Polymers Seniorcare2share

Difference Between Addition Polymerization And Condensation Polymerization Compare The Difference Between Similar Terms
Difference Between Polymer And Macromolecule Definition Classification General Properties And Differences
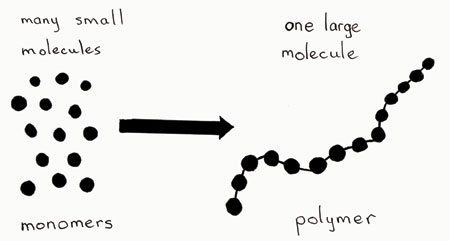
What Is The Difference Between Monomers And Polymers Socratic

What Is The Difference Between Polymers And Macromolecules Youtube

Difference Between Polymer And Macromolecule Compare The Difference Between Similar Terms

Difference Between Addition And Radical Polymerization Compare The Difference Between Similar Terms

Difference Between Polymer And Monomer Compare The Difference Between Similar Terms
Difference Between Monomer And Polymer
Difference Between Nylon And Polyester Definition Properties Uses
Difference Between Natural And Synthetic Polymers Definition Occurrence Examples

Difference Between Polymer And Monomer Compare The Difference Between Similar Terms

Question What Is The Difference Between Monomers And Polymers Seniorcare2share
Comments
Post a Comment